News
To commit
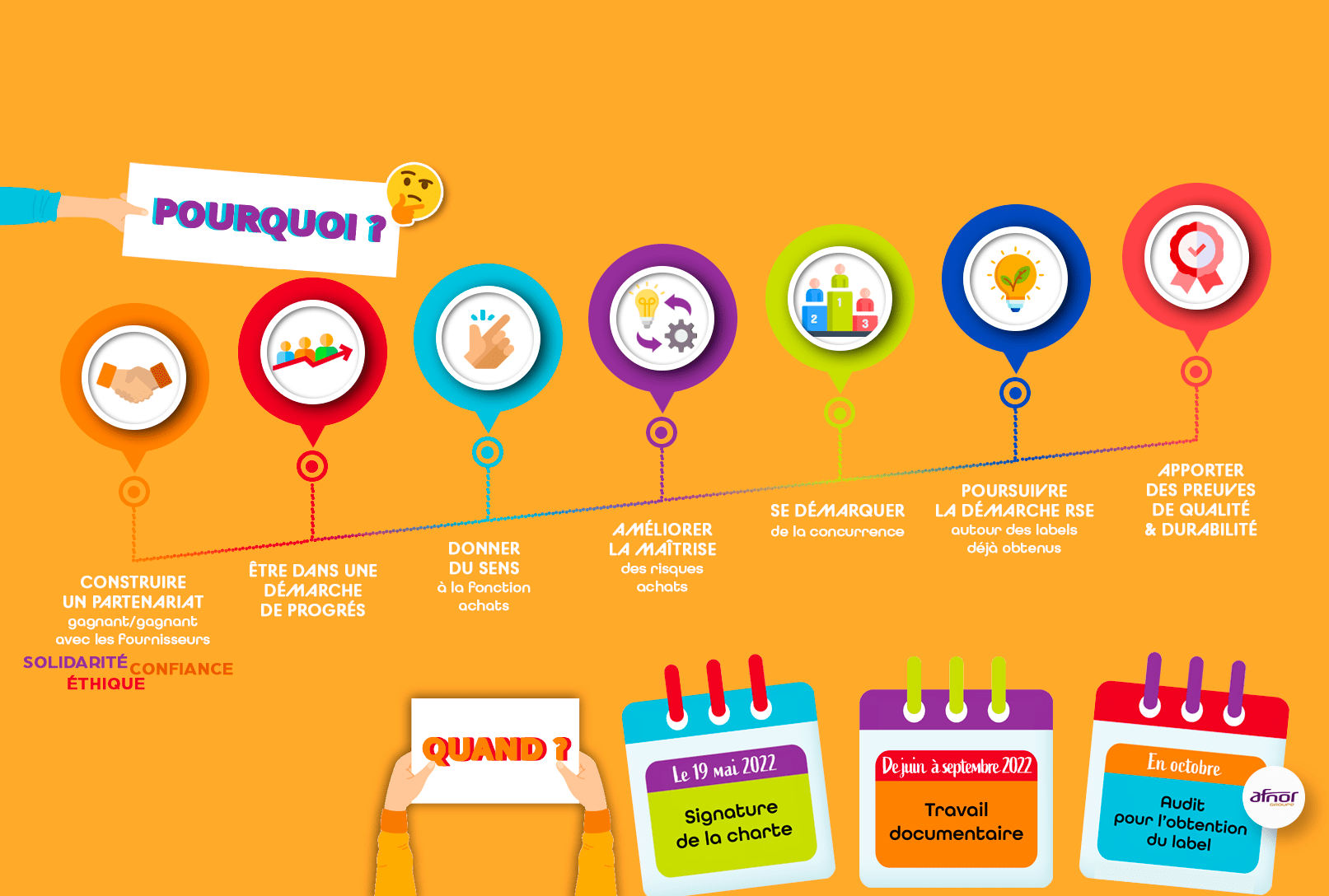
On May 19, 2022, Ingredia made a commitment to balanced supplier relations and responsible purchasing by signing the MDE and CNA Supplier Relations and Responsible Purchasing Charter.To explore
Ingredia and all the stakeholders, are committed to the process of obtaining the Responsible Supplier Relations and Purchasing label, backed by the ISO 20400 standard.To progress
Through this commitment we are inscribing our desire to anchor within our CSR policy the challenges of responsible purchasing and to establish sustainable relationships with our supplier partners. The supplier relationship is at the heart of our group's concerns and plays a crucial role in its continuous improvement and evolution.Access to the charter here!
For more information, contact us!


